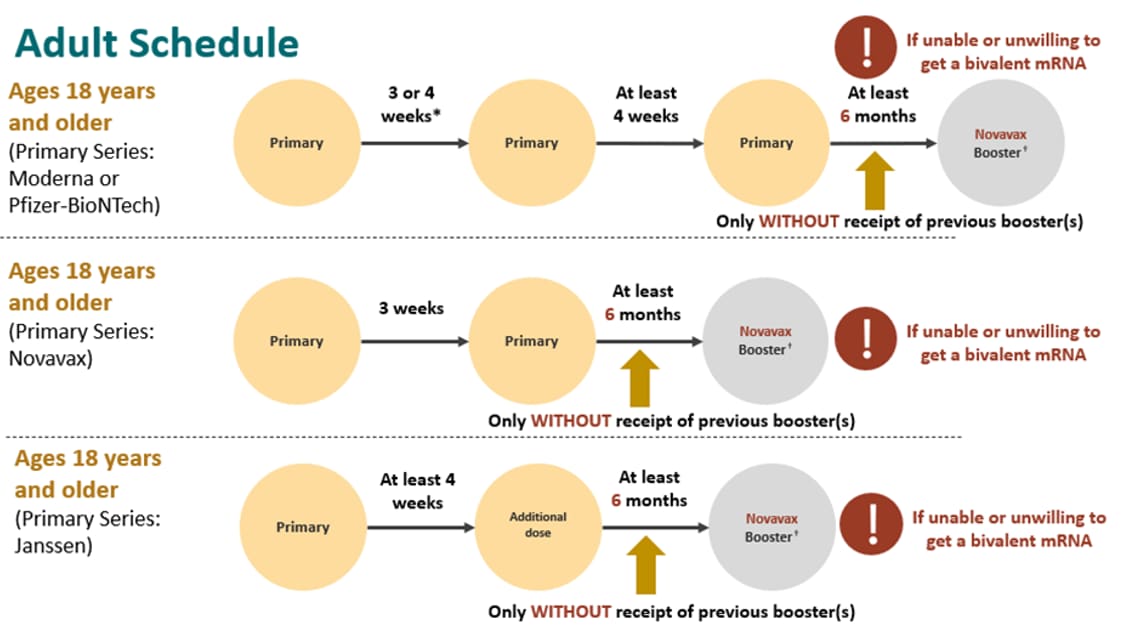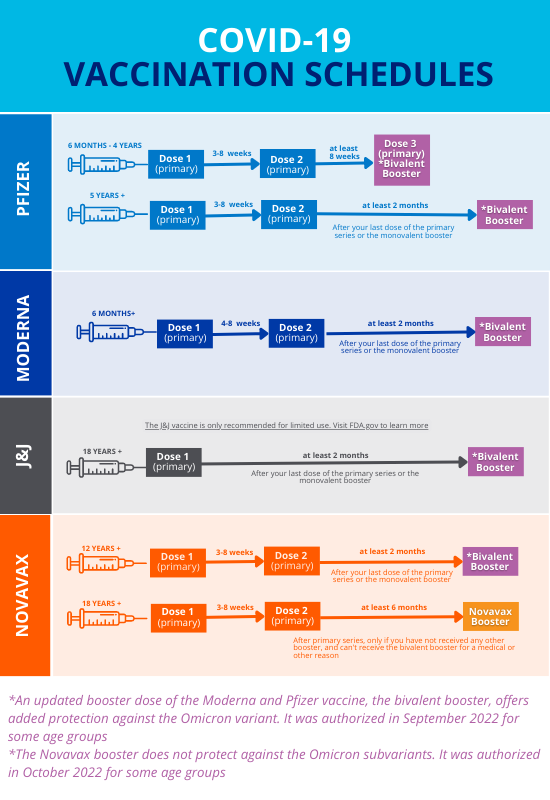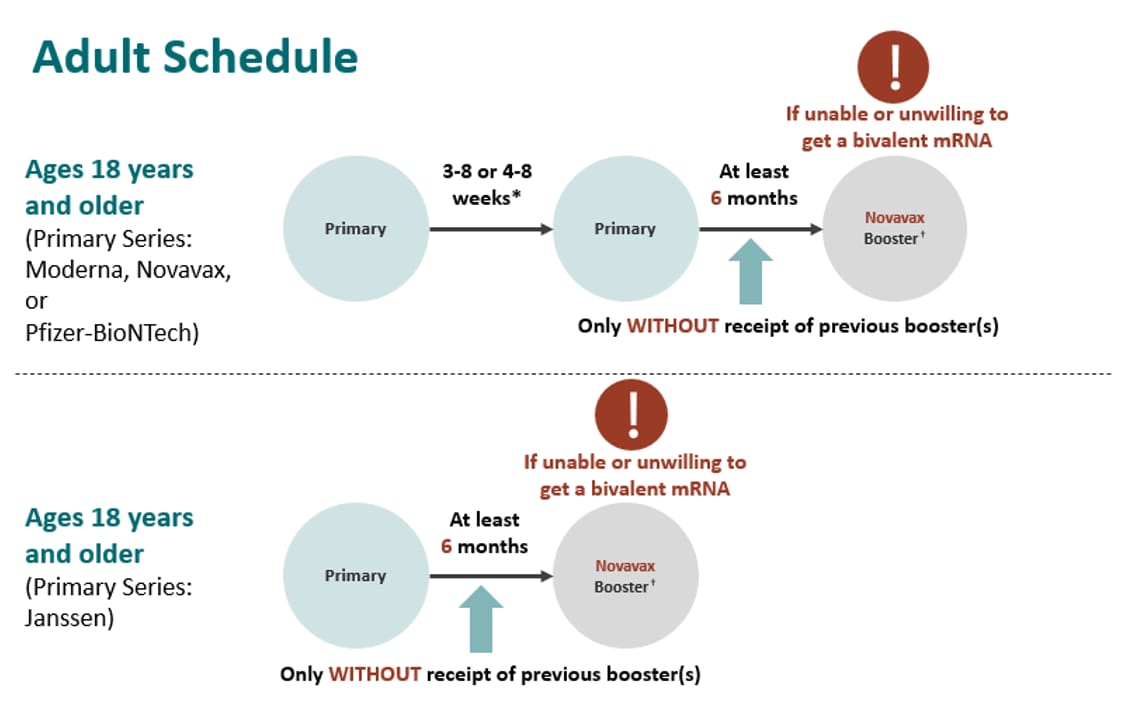
ACIP Evidence to Recommendations (EtR) for Use of Novavax COVID-19 Vaccine Booster Dose for adults ages 18 years and older under an Emergency Use Authorization | CDC
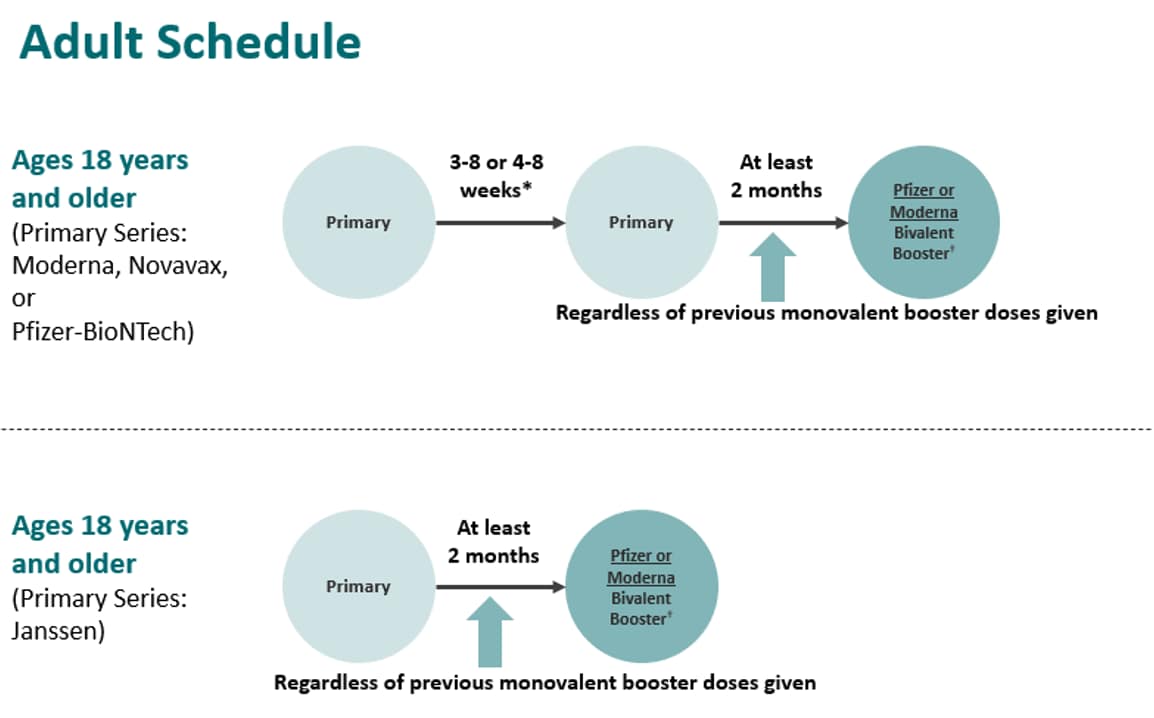
ACIP Evidence to Recommendations (EtR) for Use of Novavax COVID-19 Vaccine Booster Dose for adults ages 18 years and older under an Emergency Use Authorization | CDC

Novavax Prepared to Deliver Protein-based Monovalent XBB COVID Vaccine Consistent with FDA VRBPAC Recommendation for the Fall - Jun 15, 2023